 Index
Index

Cellulose is a polymer of β-D-Glucose, which in contrast to starch, is oriented with -CH2OH groups alternating above and below the plane of the cellulose molecule thus producing long, unbranched chains. The absence of side chains allows cellulose molecules to lie close together and form rigid structures. Cellulose is the major structural material of plants. Wood is largely cellulose, and cotton is almost pure cellulose. Cellulose can be hydrolyzed to its constituent glucose units by microorganisms that inhabit the digestive tract of termites and ruminants.
Cellulose may be modified in the laboratory by treating it with nitric acid (HNO3) to replace all the hydroxyl groups with nitrate groups (-ONO2) to produce cellulose nitrate (nitrocellulose or guncotton) which is an explosive component of smokeless powder. Partially nitrated cellulose, known as pyroxylin, is used in the manufacture of collodion, plastics, lacquers, and nail polish.

Cellulose Gum or Carboxymethyl Cellulose (CMC) is a chemical derivative of cellulose where some of the hydroxyl groups (-OH) are substituted with carboxymethyl groups (-CH2COOH). The properties of cellulose gum depend on the degree of substitution and the length of the cellulose chains. The degree of substitution (DS) is the number of carboxymethyl groups per glucose unit and may vary in commercial products from 0.4 to 1.5. Cellulose gum is non-toxic and becomes very viscous when combined with water. It is used as a thickener for foods and as an emulsion stabilizer in products like ice cream. Cellulose gum is also used in personal lubricants, diet pills, water-based paints, detergents and paper coatings.
The term "hemicellulose" is applied to the polysaccharide components of plant cell walls other than cellulose, or to polysaccharides in plant cell walls which are extractable by dilute alkaline solutions. Hemicelluloses comprise almost one-third of the carbohydrates in woody plant tissue. The chemical structure of hemicelluloses consists of long chains of a variety of pentoses, hexoses, and their corresponding uronic acids. Hemicelluloses may be found in fruit, plant stems, and grain hulls. Although hemicelluloses are not digestible, they can be fermented by yeasts and bacteria. The polysaccharides yielding pentoses on hydrolysis are called pentosans. Xylan is an example of a pentosan consisting of D-xylose units with 1β→4 linkages.
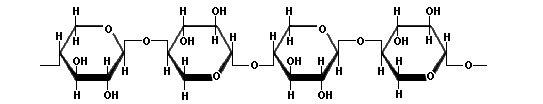
|
| Xylan |
Arabinoxylans are polysaccharides found in the bran of grasses and grains such as wheat, rye, and barley. Arabinoxylans consist of a xylan backbone with L-arabinofuranose (L-arabinose in its 5-atom ring form) attached randomly by 1α→2 and/or 1α→3 linkages to the xylose units throughout the chain. Since xylose and arabinose are both pentoses, arabinoxylans are usually classified as pentosans. Arabinoxylans are important in the baking industry. The arabinose units bind water and produce viscous compounds that affect the consistency of dough, the retention of gas bubbles from fermentation in gluten-starch films, and the final texture of baked products.

|
| Arabinoxylan |
Chitin is an unbranched polymer of N-Acetyl-D-glucosamine. It is found in fungi and is the principal component of arthropod and lower animal exoskeletons, e.g., insect, crab, and shrimp shells. It may be regarded as a derivative of cellulose, in which the hydroxyl groups of the second carbon of each glucose unit have been replaced with acetamido (-NH(C=O)CH3) groups.

|
| Chitin |
Beta-glucans consist of linear unbranched polysaccharides of β-D-Glucose like cellulose, but with one 1β→3 linkage for every three or four 1β→4 linkages. Beta-glucans form long cylindrical molecules containing up to about 250,000 glucose units. Beta-glucans occur in the bran of grains such as barley and oats, and they are recognized as being beneficial for reducing heart disease by lowering cholesterol and reducing the glycemic response. They are used comercially to modify food texture and as fat substitutes.

|
| Beta-Glucan |
Glycosaminoglycans are found in the lubricating fluid of the joints and as components of cartilage, synovial fluid, vitreous humor, bone, and heart valves. Glycosaminoglycans are long unbranched polysaccharides containing repeating disaccharide units that contain either of two amino sugar compounds -- N-acetylgalactosamine or N-acetylglucosamine, and a uronic acid such as glucuronate (glucose where carbon six forms a carboxyl group). Glycosaminoglycans are negatively charged, highly viscous molecules sometimes called mucopolysaccharides. The physiologically most important glycosaminoglycans are hyaluronic acid, dermatan sulfate, chondroitin sulfate, heparin, heparan sulfate, and keratan sulfate. Chondroitin sulfate is composed of β-D-glucuronate linked to the third carbon of N-acetylgalactosamine-4-sulfate as illustrated here. Heparin is a complex mixture of linear polysaccharides that have anticoagulant properties and vary in the degree of sulfation of the saccharide units.


Agar (agar agar) is extracted from seaweed and is used in many foods as a gelling agent. Agar is a polymer of agarobiose, a disaccharide composed of D-galactose and 3,6-anhydro-L-galactose. Highly refined agar is used as a medium for culturing bacteria, cellular tissues, and for DNA fingerprinting. Agar is used as an ingredient in desserts in Japan and other Asian countries. The gels produced with agar have a crispier texture than the desserts made with animal gelatin.
Carrageenan is a generic term for several polysaccharides also extracted from seaweed. Carrageenan compounds differ from agar in that they have sulfate groups (-OSO3-) in place of some hydroxyl groups. Carrageenan is also used for thickening, suspending, and gelling food products.

Alginate is extracted from seaweeds, such as giant kelp (Macrocystis pyrifera). The chemical constituents of alginate are random sequences of chains of β-D-mannuronic and α-L-guluronic acids attached with 1→4 linkages. Alginates are insoluble in water, but absorb water readily. They are useful as gelling and thickening agents. Alginates are used in the manufacture of textiles, paper, and cosmetics. The sodium salt of alginic acid, sodium alginate, is used in the food industry to increase viscosity and as an emulsifier. Alginates are found in food products such as ice cream and in slimming aids where they serve as appetite suppresants. In dentistry, alginates are used to make dental impressions.

Galactomannans are polysaccharides consisting of a mannose backbone with galactose side groups. The mannopyranose units are linked with 1β→4 linkages to which galactopyranose units are attached with 1α→6 linkages. Galactomannans are present in several vegetable gums that are used to increase the viscosity of food products. These are the approximate ratios of mannose to galactose for the following gums:
Guar is a legume that has been traditionally cultivated as livestock feed. Guar gum is also known by the name cyamopsis tetragonoloba which is the Latin taxonomy for the guar bean or cluster bean. Guar gum is the ground endosperm of the seeds. Approximately 85% of guar gum is guaran, a water soluble polysaccharide consisting of linear chains of mannose with 1β→4 linkages to which galactose units are attached with 1α→6 linkages. The ratio of mannose to galactose is 2:1. Guar gum has five to eight times the thickening power of starch and has many uses in the pharmaceutical industry, as a food stabilizer, and as a source of dietary fiber.

Pectin is a polysaccharide that acts as a cementing material in the cell walls of all plant tissues. The white portion of the rind of lemons and oranges contains approximately 30% pectin. Pectin is the methylated ester of polygalacturonic acid, which consists of chains of 300 to 1000 galacturonic acid units joined with 1α→4 linkages. The Degree of Esterification (DE) affects the gelling properties of pectin. The structure shown here has three methyl ester forms (-COOCH3) for every two carboxyl groups (-COOH), hence it is has a 60% degree of esterification, normally called a DE-60 pectin. Pectin is an important ingredient of fruit preserves, jellies, and jams.

Xanthan gum is a polysaccharide with a β-D-glucose backbone like cellulose, but every second glucose unit is attached to a trisaccharide consisting of mannose, glucuronic acid, and mannose. The mannose closest to the backbone has an acetic acid ester on carbon 6, and the mannose at the end of the trisaccharide is linked through carbons 6 and 4 to the second carbon of pyruvic acid. Xanthan Gum is produced by the bacterium Xanthomonas campestris, which is found on cruciferous vegetables such as cabbage and cauliflower. The negatively charged carboxyl groups on the side chains cause the molecules to form very viscous fluids when mixed with water. Xanthan gum is used as a thickener for sauces, to prevent ice crystal formation in ice cream, and as a low-calorie substitute for fat. Xanthan gum is frequently mixed with guar gum because the viscosity of the combination is greater than when either one is used alone.
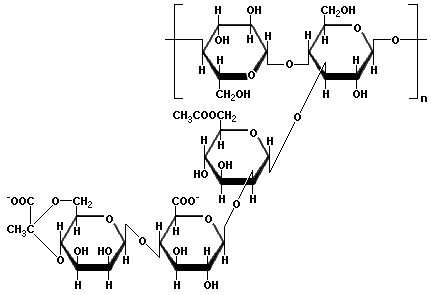
Glucomannan is a dietary fiber obtained from tubers of Amorphophallus konjac cultivated in Asia. Flour from the konjac tubers is used to make Japanese shirataki noodles, also called konnyaku noodles, which are very low in calories. Glucomannan is used as a hunger suppressant because it produces a feeling of fullness by creating very viscous solutions that retard absorption of the nutrients in food. One gram of this soluble polysaccharide can absorb up to 200 ml of water, so it is also used for absorbent articles such as disposable diapers and sanitary napkins. The polysaccharide consists of glucose (G) and mannose (M) in a proportion of 5:8 joined by 1β→4 linkages. The basic polymeric repeating unit has the pattern: GGMMGMMMMMGGM. Short side chains of 11-16 monosaccharides occur at intervals of 50-60 units of the main chain attached by 1β→3 linkages. Also, acetate groups on carbon 6 occur at every 9-19 units of the main chain. Hydrolysis of the acetate groups favors the formation of intermolecular hydrogen bonds that are responsible for the gelling action.
